 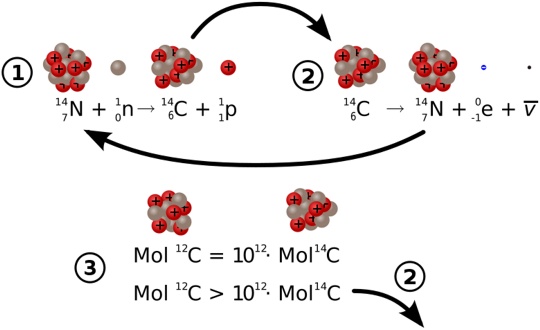
The formation of this variant happens because of a nuclear reaction between three helium nuclei called alpha particles. Scientists call it an element that provides life to the whole world. The existence of this element is very important as our earth is sort of dependent upon it. Accordingly, it has six neutrons and six protons. 
It happens to be stable because of the equal numbers of protons and neutrons in it. It is found in 99% of all Carbon on earth, hence is widely available. What is Carbon 12?Ĭarbon 12 is one of the two stable isotopes of the element Carbon. Used for replacing non-radioactive actions. Uses Used to measure the atomic mass of other nuclides. Its main source is cosmic ray reaction on nitrogen on earth. Generated in the upper layer of troposphere and stratosphere by a reaction in which thermal neutrons are absorbed by atoms of nitrogen. 
Origin Created by a nuclear fusion of three alpha particles. It makes up around 1% of the total carbon on earth. Availability It makes up approximately 90% of the total carbon on earth. It has an atomic mass of 14.0032420 u and consists of six neutrons and eight protons. Chemical Composition It has an atomic mass of 12.011 AMU and encompasses six neutrons, six protons, and six electrons. Carbon 14 is another isotope of Carbon and is generally not present in many organisms. Comparison Table Parameters of Comparison Carbon 12 Carbon 14 Definition Carbon 12 is an isotope of the element Carbon and is widely present in living organisms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
